Efficacy and safety profile in patients ≥8 years with Stage 2 T1D


TRIALNET-10 (TN-10) STUDY DESIGN
A phase 2, randomized, double-blind, event-driven, placebo-controlled study in 76 patients, 8-49 years of age with Stage 2 T1D1,2*
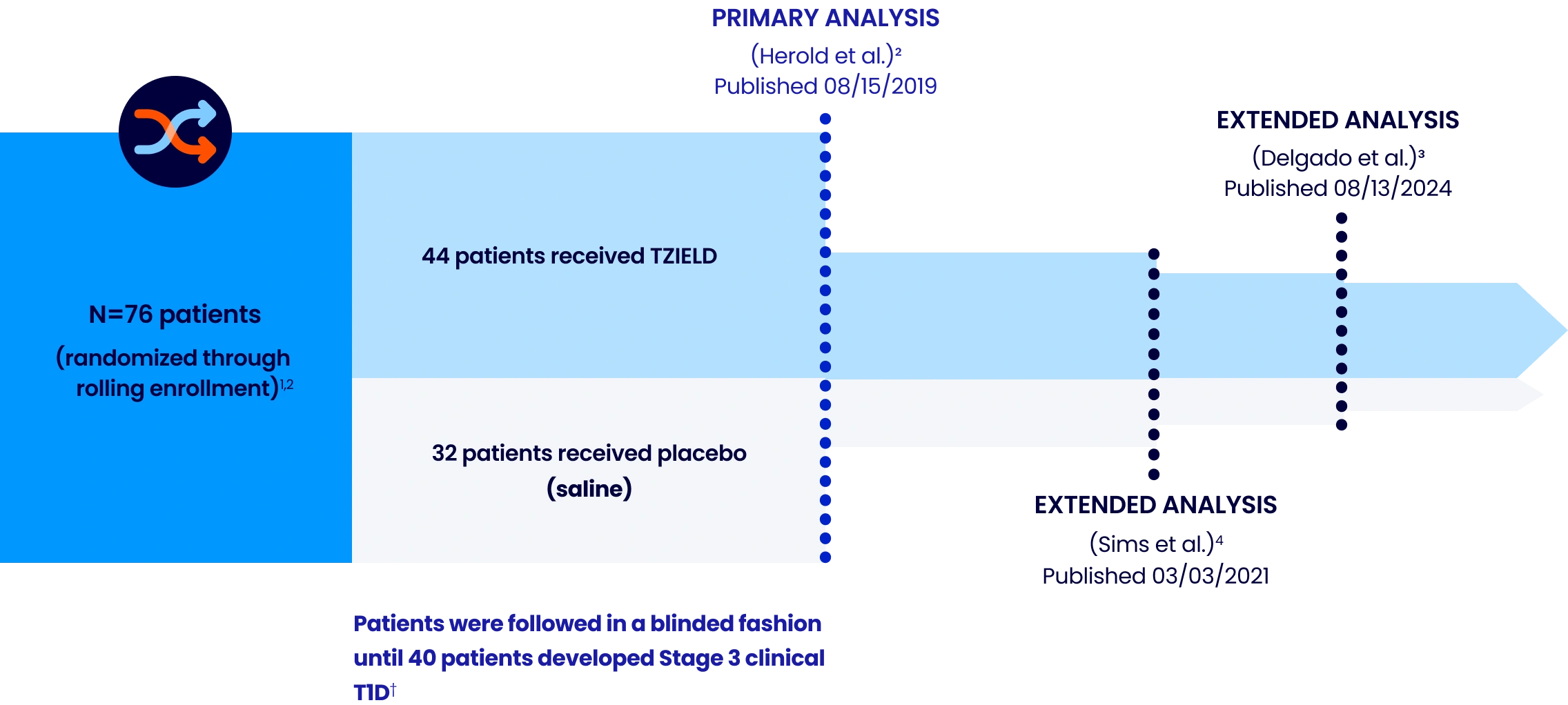
Extended analysis limitations: These extended analyses are not contained in the Prescribing Information. The TN-10 study was relatively small at the start of the study, and patient numbers decreased throughout the follow-up analyses. Therefore, definitive conclusions cannot be derived from these data.1,4
Patient results may vary.
*Stage 2 is defined as having 2 or more pancreatic islet AAbs (GADA, IAA, ICA, IA-2A, and ZnT8A) and dysglycemia on OGTT.1
†The primary efficacy endpoint was the time from randomization to diagnosis of Stage 3 clinical T1D. Criteria for Stage 3 clinical T1D onset were based on glucose testing or the presence of unequivocal hyperglycemia with acute metabolic decompensation, such as diabetic ketoacidosis.5
AAbs=autoantibodies; GADA=glutamic acid decarboxylase 65 AAb; IAA=insulin AAb; IA-2A=insulinoma-associated antigen 2 AAb; ICA=islet cell AAb; OGTT=oral glucose tolerance test; T1D=type 1 diabetes; ZnT8A=zinc transporter-8 AAb.
IN PATIENTS 8 YEARS AND OLDER WITH STAGE 2 T1D
For the first time ever, the onset of Stage 3 T1D can be delayed1,2
In the TN-10 study1,2:


more time
in presymptomatic Stage 2 vs placebo
Median time to Stage 3 onset:
TZIELD: 50 months
n=44
Placebo: 25 months
n=32
(HR 0.41; 95% CI, 0.22-0.78; P=0.0066 by an adjusted Cox proportional-hazards model stratified by age and OGTT status at randomization.)1,2
In patients with Stage 2 T1D, the median follow-up time was 51 months (range: 74 days to 2683 days).1,2*
Patients results may vary.
*Median follow-up time from the TZIELD Prescribing Information was calculated using the reverse Kaplan–Meier method.1,2
CI=confidence interval; HR=hazard ratio.
PRIMARY AND EXTENDED ANALYSIS
Median time to Stage 3 T1D onset across TN-10 analyses
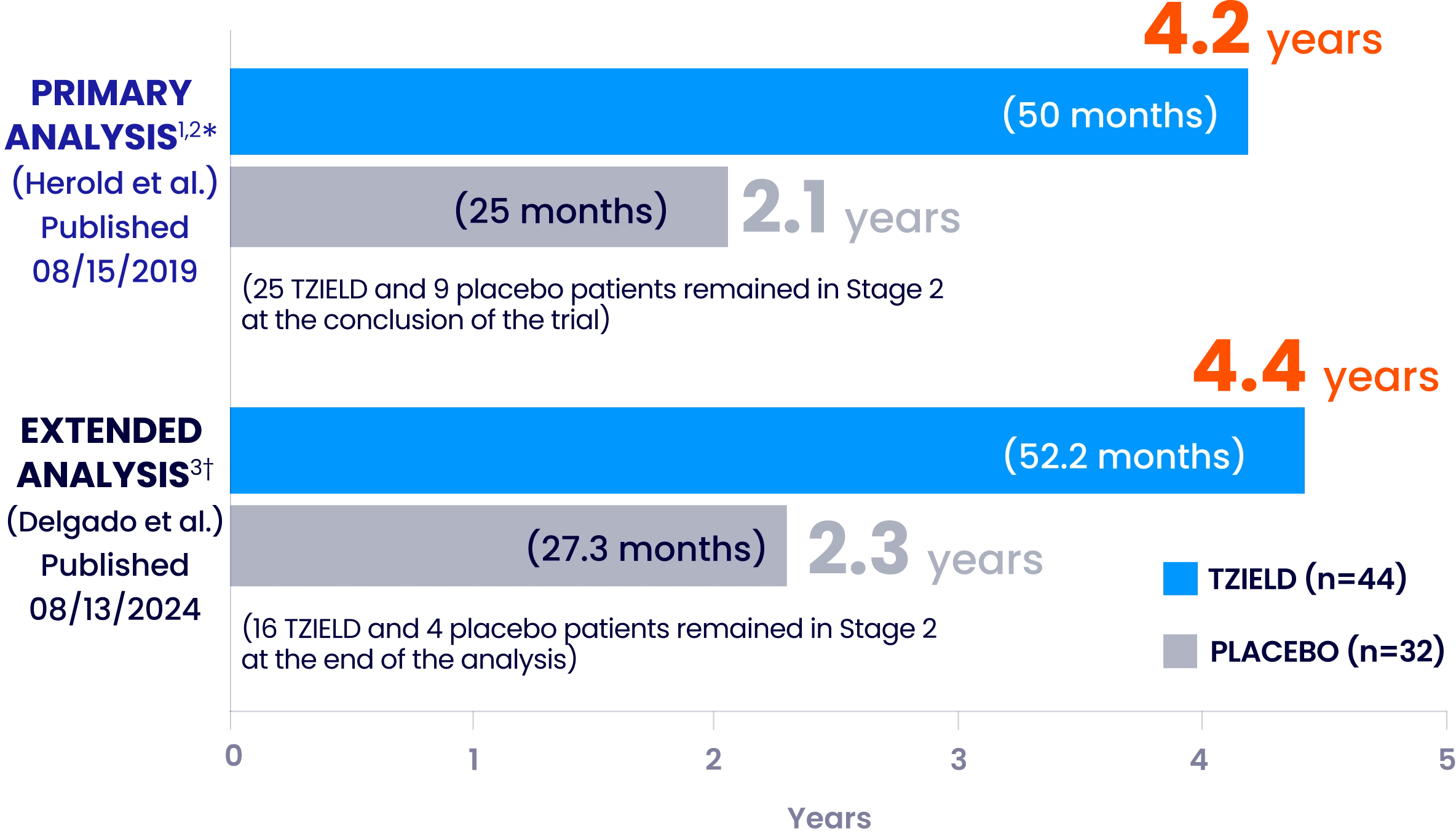
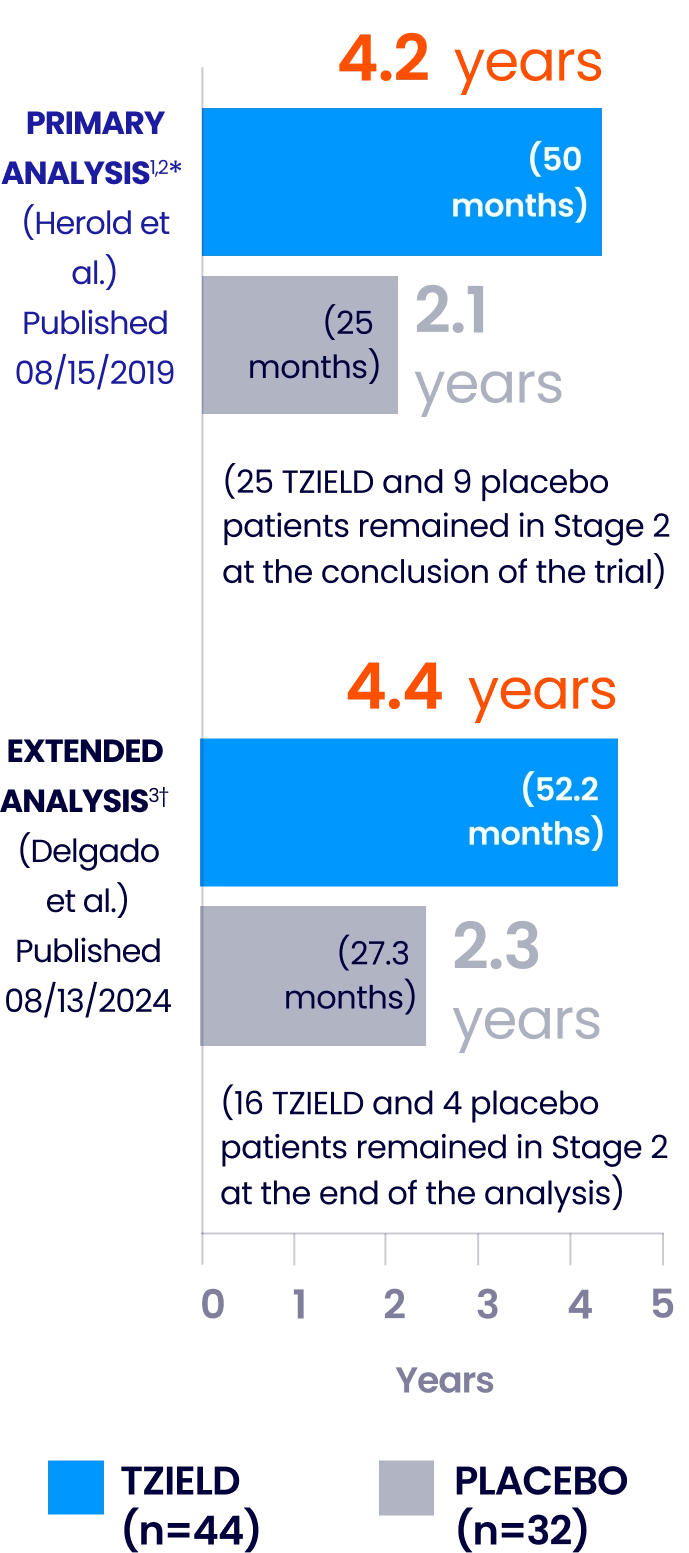
Extended analysis limitations: this extended analysis is not contained in the Prescribing Information. The TN-10 study was relatively small at the start of the study, and patient numbers decreased throughout the follow-up analyses. Therefore, definitive conclusions cannot be derived from these data.1,4
Patient results may vary.
*HR 0.41; 95% CI, 0.22-0.78; P=0.0066 by an adjusted Cox proportional-hazards model stratified by age and OGTT status at randomization. The median follow-up time was 51 months (range: 74 days to 2683 days), calculated using the reverse Kaplan-Meier method.1,2
†The median follow-up time was 80.46 months, calculated using time to T1D diagnosis, end of study participation, or administration cutoff (end of study).3
EXTENDED ANALYSIS
~4 out of 10 TZIELD-treated patients remained in Stage 2 T1D beyond 5 years3,6*
(Delgado et al. extended analysis, as of data cutoff July 2023)
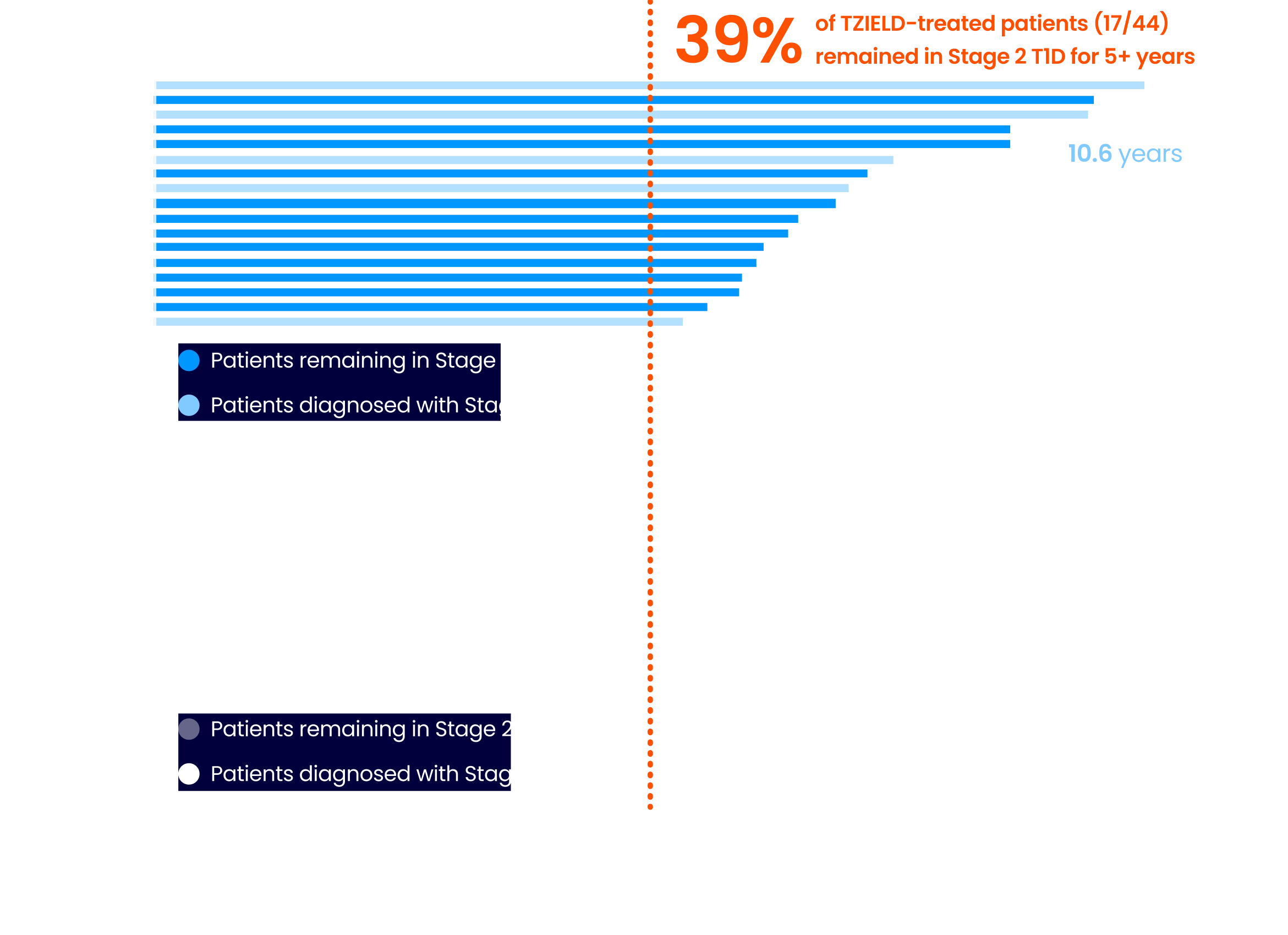
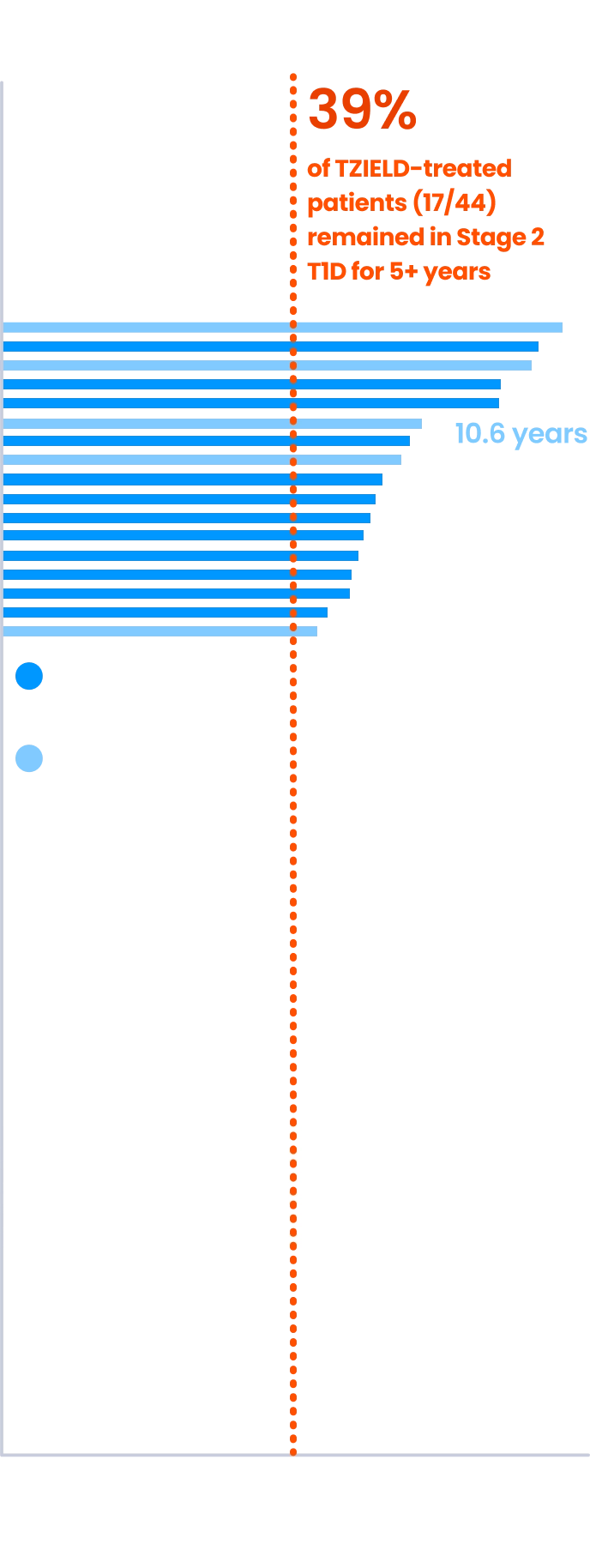
Extended analysis limitations: this extended analysis is not contained in the Prescribing Information. The TN-10 study was relatively small at the start of the study, and patient numbers decreased throughout the follow-up analyses. Therefore, definitive conclusions cannot be derived from these data.1,4
Patient results may vary.
*The median follow-up time was 80.46 months, calculated using time to TID diagnosis, end of study participation, or administration cutoff (end of study).3
TZIELD is an immunomodulator with a well-established safety profile1*
Serious adverse reactions reported throughout the TN-10 study with greater frequency in TZIELD-treated patients vs placebo-treated patients1
| Adverse reaction | TZIELD (n=44) | Placebo (n=32) |
|---|---|---|
| Cytokine release syndrome (CRS) | 2% | 0% |
| Serious infections† | 9% | 0% |
| Lymphopenia | 73% | 6% |
| Hypersensitivity reactions and serum sickness | 2% | 0% |
*Adverse reactions in TZIELD-treated patients were also evaluated in a larger pool of adult and pediatric patients who participated in 7 controlled clinical studies (including the TN-10 study).1
†Serious infections included cellulitis, gastroenteritis, pneumonia, and wound infection any time during or after the first dose of study treatment.1
Common adverse reactions (≥5%) in the TN-10 study1‡§
| Adverse reaction | TZIELD (n=44) | Placebo (n=32) |
|---|---|---|
| Lymphopenia | 73% | 6% |
| RashII | 36% | 0% |
| Leukopenia | 21% | 0% |
| Headache | 11% | 6% |
| Neutropenia | 7% | 3% |
| Increased alanine aminotransferase | 5% | 3% |
| Nausea | 5% | 3% |
| Diarrhea | 5% | 0% |
| Nasopharyngitis | 5% | 0% |
The approval of TZIELD to treat patients with Stage 2 T1D was supported by a pooled safety analysis spanning 5 clinical studies including 773 patients1:
- 44 patients with Stage 2 T1D and 729 patients from unapproved populations||
‡Adverse reactions that occurred in 2 or more TZIELD-treated patients.1
§Adverse reactions that occurred during treatment and through 28 days after the last study drug administration.1
∥Composite of rash-related terms, including rash erythematous, rash macular, rash papular, rash maculo-papular, and rash pruritic.1
¶In these studies, 436 patients received a 14-day dosing regimen of TZIELD with total drug exposure comparable to the recommended approved dosage (Study TN-10), 168 patients received a 14-day course of TZIELD with a lower total TZIELD drug exposure, and 169 patients received a 6-day course of TZIELD with a lower total TZIELD drug exposure.1
The approval of TZIELD to treat patients with Stage 2 T1D was supported by a pooled safety analysis spanning 5 clinical studies including 773 patients1:
- 44 patients with Stage 2 T1D and 729 patients from unapproved populations||
CYTOKINE RELEASE SYNDROME (CRS)
In the TN-10 study, CRS was reported in TZIELD-treated patients1
- CRS manifestations in TZIELD-treated patients included fever, nausea (with or without vomiting), fatigue, headache, myalgia, arthralgia, increased ALT, increased AST, and increased total bilirubin
- These manifestations typically occurred during the first 5 days of TZIELD treatment
Mitigation guidance for CRS1
- Premedicate with antipyretics, antihistamines and/or antiemetics prior to TZIELD treatment
- Monitor liver enzymes and bilirubin during treatment. Discontinue TZIELD treatment in patients who develop elevated ALT or AST more than 5 times the upper limit of normal (ULN) or bilirubin more than 3 times ULN
- Treat symptoms of CRS in TZIELD-treated patients with antipyretics, antihistamines and/or antiemetics
- If severe CRS develops, consider:
- Temporarily pausing TZIELD dosing for 1-2 days and if symptoms have resolved or significantly improved, administer the remaining doses on consecutive days to complete the full 14-day course or
- Discontinuing TZIELD
LYMPHOPENIA PREVALENCE IN TN-10
Lymphopenia is an expected and common adverse reaction to TZIELD that is often resolved by Week 61
Week 6: average lymphocyte counts return to baseline1,2#
Lymphopenia and treatment with TZIELD1
- For most TZIELD-treated patients who experienced lymphopenia, lymphocyte levels began to recover after the fifth day of treatment and returned to pre-treatment values within two weeks after treatment completion and without dose interruption
- Lymphopenia occurred in the absence of T-cell depletion
#Adapted from Herold KC, et al. Means and confidence intervals are shown.
Mitigation guidance for Lymphopenia1:
- Monitor lymphocyte count regularly (every 2-3 days) during TZIELD infusion and monitor for lymphocyte recovery following completion of TZIELD
- Obtain a CBC prior to starting TZIELD and monitor white blood cell counts during TZIELD treatment
- If prolonged severe lymphopenia (<500 cells per mcL lasting 1 week or longer) develops, permanently discontinue TZIELD
Additional warnings and precautions

Serious infections1
Bacterial and viral infections have occurred in TZIELD-treated patients. In clinical trials, TZIELD-treated patients had a higher rate of serious infections than control-treated patients, including gastroenteritis, cellulitis, pneumonia, abscess, sepsis. Use of TZIELD is not recommended in patients with active serious infection, or chronic infection other than localized skin infections. Monitor patients for signs and symptoms of infection during and after TZIELD treatment. If serious infection develops, treat appropriately, and discontinue TZIELD.

Vaccinations1
The safety of immunization with live-attenuated vaccines in TZIELD-treated patients has not been studied. Additionally, TZIELD may interfere with the immune response to vaccination and decrease vaccine efficacy.

Hypersensitivity reactions1
If severe hypersensitivity reactions occur, discontinue TZIELD and treat promptly. Acute hypersensitivity reactions, including serum sickness, angioedema, urticaria, rash, vomiting and bronchospasm, occurred in TZIELD-treated patients.

Overall, the safety profile of TZIELD observed in pediatric patients less than 8 years of age with Stage 2 T1D was consistent with that observed in patients 8 years of age and older with Stage 2 T1D1
Important Safety Information Anchor
IMPORTANT SAFETY INFORMATION
WARNING: Viral Reactivation
- Serious, life-threatening cases of viral reactivation, including Epstein-Barr virus (EBV) and cytomegalovirus (CMV) reactivation have been reported with TZIELD. Patients who are immunocompromised are at increased risk. The majority of serious cases occurred in patients who continued TZIELD treatment despite persistent, severe lymphopenia.
- Test patients for active EBV and CMV infection prior to starting treatment. TZIELD is not recommended in patients with laboratory or clinical evidence of active EBV or CMV infection. Adhere to lymphocyte count monitoring requirements and discontinuation recommendations. Monitor patients for signs and symptoms of viral reactivation following TZIELD treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD.
WARNINGS AND PRECAUTIONS
Viral Reactivation: Serious, life-threatening cases of viral reactivation, including EBV and CMV have been reported with TZIELD. During and within 2 months of TZIELD treatment, if primary infection or reactivation of EBV or CMV occurs, it may present with increased severity, including EBV-associated lymphoproliferative disease and organ failure. Patients who are immunocompromised, including patients with Down syndrome, may be at increased risk. The majority of serious viral reactivation cases occurred in patients who continued TZIELD despite persistent, severe lymphopenia. Prior to initiating treatment with TZIELD, evaluate patients for active EBV and CMV infection and confirm undetectable viral load (e.g., PCR testing). TZIELD is not recommended in patients with laboratory or clinical evidence of active EBV or CMV infection. During treatment with TZIELD, regularly monitor lymphocyte counts and monitor patients for signs and symptoms of viral reactivation during treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD and obtain viral load (e.g., PCR) promptly. If viral reactivation is confirmed, permanently discontinue TZIELD.
Cytokine Release Syndrome (CRS): CRS occurred in TZIELD-treated patients during the treatment period and through 28 days after the last drug administration. CRS manifestations in TZIELD-treated patients included fever, nausea (with or without vomiting), fatigue, headache, myalgia, arthralgia, increased ALT, increased AST, and increased total bilirubin. These manifestations typically occurred during the first 5 days of TZIELD treatment. Prior to TZIELD treatment, premedicate with antipyretics, antihistamines and/or antiemetics, and treat similarly if symptoms occur during treatment. If severe CRS develops, consider pausing dosing for 1 day to 2 days and administering the remaining doses to complete the full 14-day course on consecutive days; or discontinue treatment. Monitor liver enzymes during treatment. Discontinue TZIELD treatment in patients who develop elevated alanine aminotransferase or aspartate aminotransferase more than 5 times the upper limit of normal (ULN) or bilirubin more than 3 times ULN.
Serious Infections: Use of TZIELD is not recommended in patients with active serious infection or chronic infection other than localized skin infections. Monitor patients for signs and symptoms of infection during and after TZIELD administration. If serious infection develops, treat appropriately, and discontinue TZIELD.
Lymphopenia: Lymphopenia occurred in most TZIELD-treated patients. For most patients, lymphocyte levels began to recover after the fifth day of treatment and returned to pretreatment values within two weeks after treatment completion and without dose interruption. Obtain a CBC prior to starting TZIELD and monitor white blood cell counts during TZIELD treatment. If prolonged severe lymphopenia develops (<500 cells per mcL lasting 1 week or longer), permanently discontinue TZIELD.
Hypersensitivity Reactions: Acute hypersensitivity reactions including serum sickness, angioedema, urticaria, rash, vomiting and bronchospasm occurred in TZIELD-treated patients. If severe hypersensitivity reactions occur, discontinue TZIELD and treat promptly.
Vaccinations: The safety of immunization with live-attenuated (live) vaccines with TZIELD-treated patients has not been studied. TZIELD may interfere with immune response to vaccination and decrease vaccine efficacy. Administer all age-appropriate vaccinations prior to starting TZIELD.
- Administer inactivated (killed) vaccines or mRNA vaccines at least 2 weeks prior to treatment. Inactivated vaccines are not recommended during treatment or 6 weeks after completion of treatment.
- Administer live vaccines at least 8 weeks prior to treatment. Live vaccines are not recommended during treatment, or up to 52 weeks after treatment.
ADVERSE REACTIONS
Most common adverse reactions were lymphopenia, vomiting, rash, leukopenia, diarrhea and headache.
USE IN SPECIFIC POPULATIONS
- Pregnancy: May cause fetal harm. To minimize exposure to a fetus, avoid use of TZIELD during pregnancy and at least 30 days prior to planned pregnancy. Report pregnancies to us at our Adverse Event reporting line at 1-800-633-1610 or visit https://ae.reporting.sanofi
- Lactation: A lactating woman may consider pumping and discarding breast milk during and for 20 days after TZIELD administration.
Please see full Prescribing Information, including Boxed WARNING and patient selection criteria.
INDICATION
TZIELD (teplizumab-mzwv) is indicated to delay the onset of Stage 3 type 1 diabetes (T1D) in adults and pediatric patients 1 year of age and older with Stage 2 T1D.
REFERENCES
- TZIELD Prescribing Information. Provention Bio, Inc.
- Herold KC, Bundy BN, Long SA, et al; Type 1 Diabetes TrialNet Study Group. An anti-CD3 antibody, teplizumab, in relatives at risk for type 1 diabetes. N Engl J Med. 2019;381(7):603-613.
- Lledó-Delgado A, Preston-Hurlburt P, Currie S, et al. Teplizumab induces persistent changes in the antigen-specific repertoire in individuals at risk for type 1 diabetes. J Clin Invest. 2024;134(18):e177492.
- Sims EK, Bundy BN, Stier K, et al; Type 1 Diabetes TrialNet Study Group. Teplizumab improves and stabilizes beta cell function in antibody-positive high-risk individuals + Supplementary Appendix. Sci Transl Med. 2021;13(583):eabc8980.
- Data on file. Sanofi.
- Lledó-Delgado A, Preston-Hurlburt P, Currie S, et al. Teplizumab induces persistent changes in the antigen-specific repertoire in individuals at risk for type 1 diabetes. J Clin Invest. 2024;134(18)(suppl):e177492.
IMPORTANT SAFETY INFORMATION
INDICATION
IMPORTANT SAFETY INFORMATION
WARNING: Viral Reactivation
- Serious, life-threatening cases of viral reactivation, including Epstein-Barr virus (EBV) and cytomegalovirus (CMV) reactivation have been reported with TZIELD. Patients who are immunocompromised are at increased risk. The majority of serious cases occurred in patients who continued TZIELD treatment despite persistent, severe lymphopenia.
- Test patients for active EBV and CMV infection prior to starting treatment. TZIELD is not recommended in patients with laboratory or clinical evidence of active EBV or CMV infection. Adhere to lymphocyte count monitoring requirements and discontinuation recommendations. Monitor patients for signs and symptoms of viral reactivation following TZIELD treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD.
WARNINGS AND PRECAUTIONS
Viral Reactivation: Serious, life-threatening cases of viral reactivation, including EBV and CMV have been reported with TZIELD. During and within 2 months of TZIELD treatment, if primary infection or reactivation of EBV or CMV occurs, it may present with increased severity, including EBV-associated lymphoproliferative disease and organ failure. Patients who are immunocompromised, including patients with Down syndrome, may be at increased risk. The majority of serious viral reactivation cases occurred in patients who continued TZIELD despite persistent, severe lymphopenia. Prior to initiating treatment with TZIELD, evaluate patients for active EBV and CMV infection and confirm undetectable viral load (e.g., PCR testing). TZIELD is not recommended in patients with laboratory or clinical evidence of active EBV or CMV infection. During treatment with TZIELD, regularly monitor lymphocyte counts and monitor patients for signs and symptoms of viral reactivation during treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD and obtain viral load (e.g., PCR) promptly. If viral reactivation is confirmed, permanently discontinue TZIELD.
Cytokine Release Syndrome (CRS): CRS occurred in TZIELD-treated patients during the treatment period and through 28 days after the last drug administration. CRS manifestations in TZIELD-treated patients included fever, nausea (with or without vomiting), fatigue, headache, myalgia, arthralgia, increased ALT, increased AST, and increased total bilirubin. These manifestations typically occurred during the first 5 days of TZIELD treatment. Prior to TZIELD treatment, premedicate with antipyretics, antihistamines and/or antiemetics, and treat similarly if symptoms occur during treatment. If severe CRS develops, consider pausing dosing for 1 day to 2 days and administering the remaining doses to complete the full 14-day course on consecutive days; or discontinue treatment. Monitor liver enzymes during treatment. Discontinue TZIELD treatment in patients who develop elevated alanine aminotransferase or aspartate aminotransferase more than 5 times the upper limit of normal (ULN) or bilirubin more than 3 times ULN.
Serious Infections: Use of TZIELD is not recommended in patients with active serious infection or chronic infection other than localized skin infections. Monitor patients for signs and symptoms of infection during and after TZIELD administration. If serious infection develops, treat appropriately, and discontinue TZIELD.
Lymphopenia: Lymphopenia occurred in most TZIELD-treated patients. For most patients, lymphocyte levels began to recover after the fifth day of treatment and returned to pretreatment values within two weeks after treatment completion and without dose interruption. Obtain a CBC prior to starting TZIELD and monitor white blood cell counts during TZIELD treatment. If prolonged severe lymphopenia develops (<500 cells per mcL lasting 1 week or longer), permanently discontinue TZIELD.
Hypersensitivity Reactions: Acute hypersensitivity reactions including serum sickness, angioedema, urticaria, rash, vomiting and bronchospasm occurred in TZIELD-treated patients. If severe hypersensitivity reactions occur, discontinue TZIELD and treat promptly.
Vaccinations: The safety of immunization with live-attenuated (live) vaccines with TZIELD-treated patients has not been studied. TZIELD may interfere with immune response to vaccination and decrease vaccine efficacy. Administer all age-appropriate vaccinations prior to starting TZIELD.
- Administer inactivated (killed) vaccines or mRNA vaccines at least 2 weeks prior to treatment. Inactivated vaccines are not recommended during treatment or 6 weeks after completion of treatment.
- Administer live vaccines at least 8 weeks prior to treatment. Live vaccines are not recommended during treatment, or up to 52 weeks after treatment.
ADVERSE REACTIONS
Most common adverse reactions were lymphopenia, vomiting, rash, leukopenia, diarrhea and headache.
USE IN SPECIFIC POPULATIONS
- Pregnancy: May cause fetal harm. To minimize exposure to a fetus, avoid use of TZIELD during pregnancy and at least 30 days prior to planned pregnancy. Report pregnancies to us at our Adverse Event reporting line at 1-800-633-1610 or visit https://ae.reporting.sanofi
- Lactation: A lactating woman may consider pumping and discarding breast milk during and for 20 days after TZIELD administration.
Please see full Prescribing Information, including Boxed WARNING and patient selection criteria.
INDICATION
TZIELD (teplizumab-mzwv) is indicated to delay the onset of Stage 3 type 1 diabetes (T1D) in adults and pediatric patients 1 year of age and older with Stage 2 T1D.



