What are the stages of T1D?
Autoimmune T1D has 4 distinct and detectable stages.1


Once diagnosed, autoimmune T1D progression is not a matter of if, but WHEN
Stages are identified by the presence of multiple autoantibodies and glycemic testing.
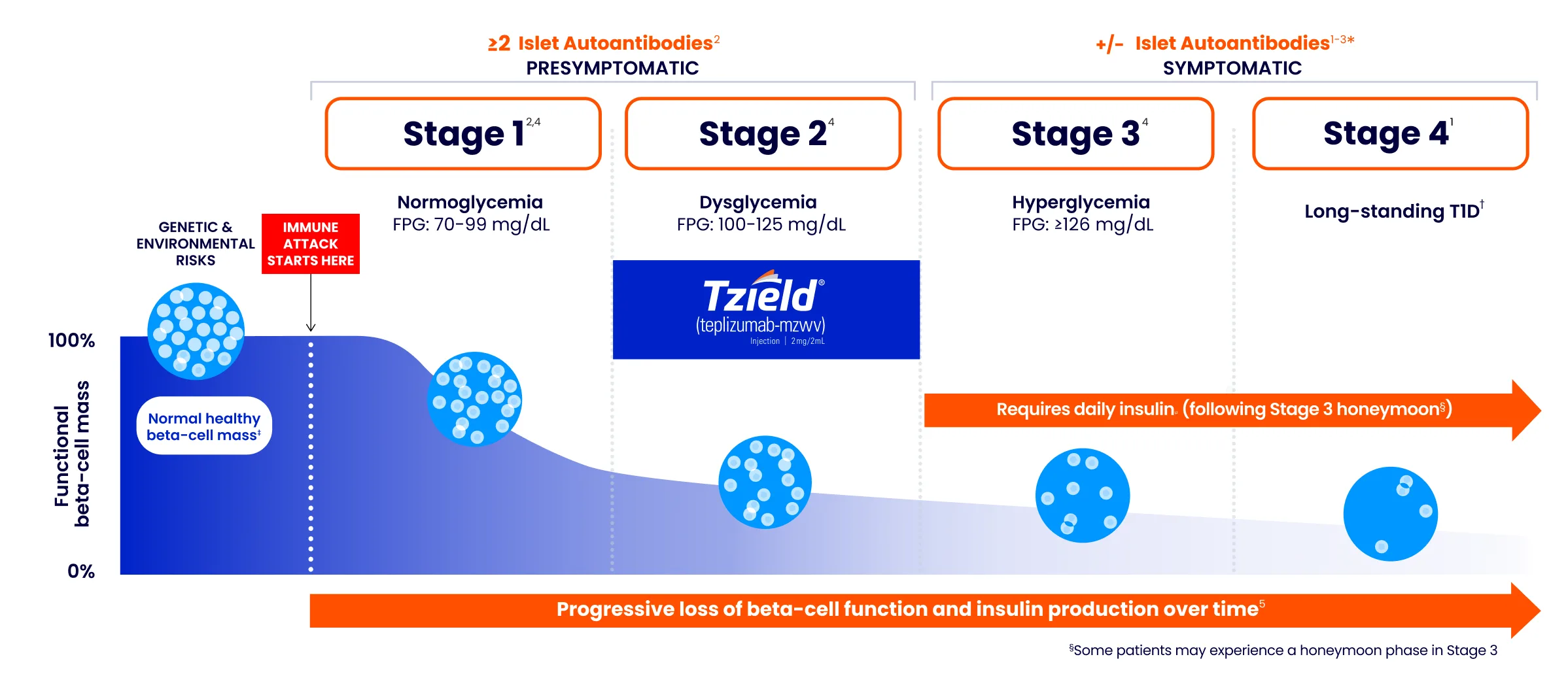
Figure adapted from Breakthrough T1D, formerly known as JDRF.1
*Autoantibodies may become absent at this stage.3
†Long-standing T1D is often defined as having T1D for at least 5 years.6,7
‡For illustrative purposes only.
If patients test positive for multiple autoantibodies, then in addition to an autoantibody test, perform routine glycemic assessment to confirm diagnosis and staging.2,4
Once patients are in Stage 1 T1D, progression to Stage 3 becomes inevitable2
Once 2 or more islet autoantibodies are present and glycemic assessment confirms diagnosis, it's not a matter of if, but WHEN a patient progresses to clinical Stage 3 T1D. Once in Stage 1 or 2, the lifetime risk for progression to Stage 3 T1D is ~100%.
Risk of progressing to Stage 3 T1D
STAGE 1
5-year risk
44%
10-year risk
70%
Lifetime risk approaches
100%
STAGE 2
5-year risk
75%
Lifetime risk approaches
100%
Younger children progress to Stage 3 T1D faster than older children and adults.2,8
Identifying autoimmune T1D early can give you more time9
Screen and stage for the potential opportunity to intervene before Stage 3 T1D.9,10

Refer your patients to specialists in a number of areas, including mental health and other support areas.

Talk to your patients and their care teams to know when and how to seek presymptomatic intervention.

Advise patients and caregivers to be vigilant for symptoms of hyperglycemia and DKA.
Per year, the average clinical symptomatic T1D patient may experience:

insulin needle injections annually11

insulin pump site changes annually12

T1D-related decisions
(ie, constant monitoring of glucose, food, and activity levels)13
Screening and monitoring reduced the risk of DKA at initial Stage 3 T1D onset by ~88% in children15
Up to 62% of newly diagnosed patients with clinical T1D without prior screening had DKA at diagnosis.16
DKA occurs when the body doesn't have enough insulin to allow blood sugar into cells for use as energy. Instead, the liver breaks down fat for fuel, a process that produces acids called ketones.17-19
DKA can have negative and long-lasting effects. A patient’s long-term glycemic management can be affected by a DKA event at diagnosis.17-19


Learn how to support your patients' treatment
Important Safety Information Anchor
IMPORTANT SAFETY INFORMATION
WARNING: Viral Reactivation
- Serious, life-threatening cases of viral reactivation, including Epstein-Barr virus (EBV) and cytomegalovirus (CMV) reactivation have been reported with TZIELD. Patients who are immunocompromised are at increased risk. The majority of serious cases occurred in patients who continued TZIELD treatment despite persistent, severe lymphopenia.
- Test patients for active EBV and CMV infection prior to starting treatment. TZIELD is not recommended in patients with laboratory or clinical evidence of active EBV or CMV infection. Adhere to lymphocyte count monitoring requirements and discontinuation recommendations. Monitor patients for signs and symptoms of viral reactivation following TZIELD treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD.
WARNINGS AND PRECAUTIONS
Viral Reactivation: Serious, life-threatening cases of viral reactivation, including EBV and CMV have been reported with TZIELD. During and within 2 months of TZIELD treatment, if primary infection or reactivation of EBV or CMV occurs, it may present with increased severity, including EBV-associated lymphoproliferative disease and organ failure. Patients who are immunocompromised, including patients with Down syndrome, may be at increased risk. The majority of serious viral reactivation cases occurred in patients who continued TZIELD despite persistent, severe lymphopenia. Prior to initiating treatment with TZIELD, evaluate patients for active EBV and CMV infection and confirm undetectable viral load (e.g., PCR testing). TZIELD is not recommended in patients with laboratory or clinical evidence of active EBV or CMV infection. During treatment with TZIELD, regularly monitor lymphocyte counts and monitor patients for signs and symptoms of viral reactivation during treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD and obtain viral load (e.g., PCR) promptly. If viral reactivation is confirmed, permanently discontinue TZIELD.
Cytokine Release Syndrome (CRS): CRS occurred in TZIELD-treated patients during the treatment period and through 28 days after the last drug administration. CRS manifestations in TZIELD-treated patients included fever, nausea (with or without vomiting), fatigue, headache, myalgia, arthralgia, increased ALT, increased AST, and increased total bilirubin. These manifestations typically occurred during the first 5 days of TZIELD treatment. Prior to TZIELD treatment, premedicate with antipyretics, antihistamines and/or antiemetics, and treat similarly if symptoms occur during treatment. If severe CRS develops, consider pausing dosing for 1 day to 2 days and administering the remaining doses to complete the full 14-day course on consecutive days; or discontinue treatment. Monitor liver enzymes during treatment. Discontinue TZIELD treatment in patients who develop elevated alanine aminotransferase or aspartate aminotransferase more than 5 times the upper limit of normal (ULN) or bilirubin more than 3 times ULN.
Serious Infections: Use of TZIELD is not recommended in patients with active serious infection or chronic infection other than localized skin infections. Monitor patients for signs and symptoms of infection during and after TZIELD administration. If serious infection develops, treat appropriately, and discontinue TZIELD.
Lymphopenia: Lymphopenia occurred in most TZIELD-treated patients. For most patients, lymphocyte levels began to recover after the fifth day of treatment and returned to pretreatment values within two weeks after treatment completion and without dose interruption. Obtain a CBC prior to starting TZIELD and monitor white blood cell counts during TZIELD treatment. If prolonged severe lymphopenia develops (<500 cells per mcL lasting 1 week or longer), permanently discontinue TZIELD.
Hypersensitivity Reactions: Acute hypersensitivity reactions including serum sickness, angioedema, urticaria, rash, vomiting and bronchospasm occurred in TZIELD-treated patients. If severe hypersensitivity reactions occur, discontinue TZIELD and treat promptly.
Vaccinations: The safety of immunization with live-attenuated (live) vaccines with TZIELD-treated patients has not been studied. TZIELD may interfere with immune response to vaccination and decrease vaccine efficacy. Administer all age-appropriate vaccinations prior to starting TZIELD.
- Administer inactivated (killed) vaccines or mRNA vaccines at least 2 weeks prior to treatment. Inactivated vaccines are not recommended during treatment or 6 weeks after completion of treatment.
- Administer live vaccines at least 8 weeks prior to treatment. Live vaccines are not recommended during treatment, or up to 52 weeks after treatment.
ADVERSE REACTIONS
Most common adverse reactions were lymphopenia, vomiting, rash, leukopenia, diarrhea and headache.
USE IN SPECIFIC POPULATIONS
- Pregnancy: May cause fetal harm. To minimize exposure to a fetus, avoid use of TZIELD during pregnancy and at least 30 days prior to planned pregnancy. Report pregnancies to us at our Adverse Event reporting line at 1-800-633-1610 or visit https://ae.reporting.sanofi
- Lactation: A lactating woman may consider pumping and discarding breast milk during and for 20 days after TZIELD administration.
Please see full Prescribing Information, including Boxed WARNING and patient selection criteria.
INDICATION
TZIELD (teplizumab-mzwv) is indicated to delay the onset of Stage 3 type 1 diabetes (T1D) in adults and pediatric patients 1 year of age and older with Stage 2 T1D.
REFERENCES
- The stages of type 1 diabetes and why they’re important. Breakthrough T1D. Accessed September 18, 2025. https://breakthrought1d.org.au/app/uploads/2025/03/Stages-of-T1D-V2.pdf
- Insel RA, Dunne JL, Atkinson MA, et al. Staging presymptomatic type 1 diabetes: a scientific statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care. 2015;38(10):1964-1974.
- American Diabetes Association Professional Practice Committee. Standards of Care in Diabetes—2026. Diabetes Care. 2026;49(suppl 1):S1-S371.
- Understanding A1C diagnosis. American Diabetes Association. Accessed April 17, 2025. https://www.diabetes.org/a1c
- Scheiner G, Weiner S, Kruger DF, et al. Screening for type 1 diabetes: role of the diabetes care and education specialist. ADCES Pract. 2022;10(5):20-25.
- Perkins BA, Lovblom LE, Lanctôt SO, et al. Discoveries from the study of longstanding type 1 diabetes. Diabetologia. 2021;64(6):1189-1200.
- Oram RA, McDonald TJ, Shields BM, et al; UNITED team. Most people with long-duration type 1 diabetes in a large population-based study are insulin microsecretors. Diabetes Care. 2015;38(2):323-328.
- Jacobsen L, Bocchino L, Evans-Molina C, et al. The risk of progression to type 1 diabetes is highly variable in individuals with multiple autoantibodies following screening. Diabetologia. 2020;63:588-596.
- TZIELD Prescribing Information. Provention Bio, Inc.
- Edelman S. Early intervention by family physicians to delay type 1 diabetes. J Fam Pract. 2023;72(6 suppl):S19-S24.
- Janež A, Guja C, Mitrakou A, et al. Insulin therapy in adults with type 1 diabetes mellitus: A narrative review. Diabetes Ther. 2020;11(2):387-409.
- Schmid V, Hohberg C, Borchert M, et al. Pilot study for assessment of optimal frequency for changing catheters in insulin pump therapy—trouble starts on day 3. J Diabetes Sci Technol. 2010;4(4):976-982
- Tack C, Lancee GJ, Heeren B, et al. Glucose control, disease burden, and educational gaps in people with type 1 diabetes: exploratory study of an integrated mobile diabetes app. JMIR Diabetes. 2018;3(4):1-13.
- Koyama AK, Hora IA, Bullard KM, et al. State-specific prevalence of depression among adults with and without diabetes — United States, 2011-2019. Prev Chronic Dis. 2023;20:E70.
- Winkler C, Schober E, Ziegler A-G, et al. Markedly reduced rate of diabetic ketoacidosis at onset of type 1 diabetes in relatives screened for islet autoantibodies. Pediatr Diabetes. 2012;13(4):308-313.
- Beliard K, Ebekozien O, Demeterco-Berggren C, et al. Increased DKA at presentation among newly diagnosed type 1 diabetes patients with or without COVID-19: data from a multi-site surveillance registry. J Diabetes. 2021;13(3):270-272.
- Diabetic ketoacidosis. Centers for Disease Control and Prevention. May 15, 2024. Accessed January 14, 2026. https://www.cdc.gov/diabetes/about/diabeticketoacidosis.pdf
- Elding Larsson H, Vehik K, Bell R, et al; TEDDY Study Group; SEARCH Study Group; Swediabkids Study Group; DPV Study Group; Finnish Diabetes Registry Study Group. Reduced prevalence of diabetic ketoacidosis at diagnosis of type 1 diabetes in young children participating in longitudinal follow-up. Diabetes Care. 2011;34(11):2347-2352.
- Bowden SA, Duck MM, Hoffman RP. Young children (<5 yr) and adolescents (>12 yr) with type 1 diabetes mellitus have low rate of partial remission: diabetic ketoacidosis is an important risk factor. Pediatr Diabetes. 2008;9(3 Pt 1):197-201.
IMPORTANT SAFETY INFORMATION
INDICATION
IMPORTANT SAFETY INFORMATION
WARNING: Viral Reactivation
- Serious, life-threatening cases of viral reactivation, including Epstein-Barr virus (EBV) and cytomegalovirus (CMV) reactivation have been reported with TZIELD. Patients who are immunocompromised are at increased risk. The majority of serious cases occurred in patients who continued TZIELD treatment despite persistent, severe lymphopenia.
- Test patients for active EBV and CMV infection prior to starting treatment. TZIELD is not recommended in patients with laboratory or clinical evidence of active EBV or CMV infection. Adhere to lymphocyte count monitoring requirements and discontinuation recommendations. Monitor patients for signs and symptoms of viral reactivation following TZIELD treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD.
WARNINGS AND PRECAUTIONS
Viral Reactivation: Serious, life-threatening cases of viral reactivation, including EBV and CMV have been reported with TZIELD. During and within 2 months of TZIELD treatment, if primary infection or reactivation of EBV or CMV occurs, it may present with increased severity, including EBV-associated lymphoproliferative disease and organ failure. Patients who are immunocompromised, including patients with Down syndrome, may be at increased risk. The majority of serious viral reactivation cases occurred in patients who continued TZIELD despite persistent, severe lymphopenia. Prior to initiating treatment with TZIELD, evaluate patients for active EBV and CMV infection and confirm undetectable viral load (e.g., PCR testing). TZIELD is not recommended in patients with laboratory or clinical evidence of active EBV or CMV infection. During treatment with TZIELD, regularly monitor lymphocyte counts and monitor patients for signs and symptoms of viral reactivation during treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD and obtain viral load (e.g., PCR) promptly. If viral reactivation is confirmed, permanently discontinue TZIELD.
Cytokine Release Syndrome (CRS): CRS occurred in TZIELD-treated patients during the treatment period and through 28 days after the last drug administration. CRS manifestations in TZIELD-treated patients included fever, nausea (with or without vomiting), fatigue, headache, myalgia, arthralgia, increased ALT, increased AST, and increased total bilirubin. These manifestations typically occurred during the first 5 days of TZIELD treatment. Prior to TZIELD treatment, premedicate with antipyretics, antihistamines and/or antiemetics, and treat similarly if symptoms occur during treatment. If severe CRS develops, consider pausing dosing for 1 day to 2 days and administering the remaining doses to complete the full 14-day course on consecutive days; or discontinue treatment. Monitor liver enzymes during treatment. Discontinue TZIELD treatment in patients who develop elevated alanine aminotransferase or aspartate aminotransferase more than 5 times the upper limit of normal (ULN) or bilirubin more than 3 times ULN.
Serious Infections: Use of TZIELD is not recommended in patients with active serious infection or chronic infection other than localized skin infections. Monitor patients for signs and symptoms of infection during and after TZIELD administration. If serious infection develops, treat appropriately, and discontinue TZIELD.
Lymphopenia: Lymphopenia occurred in most TZIELD-treated patients. For most patients, lymphocyte levels began to recover after the fifth day of treatment and returned to pretreatment values within two weeks after treatment completion and without dose interruption. Obtain a CBC prior to starting TZIELD and monitor white blood cell counts during TZIELD treatment. If prolonged severe lymphopenia develops (<500 cells per mcL lasting 1 week or longer), permanently discontinue TZIELD.
Hypersensitivity Reactions: Acute hypersensitivity reactions including serum sickness, angioedema, urticaria, rash, vomiting and bronchospasm occurred in TZIELD-treated patients. If severe hypersensitivity reactions occur, discontinue TZIELD and treat promptly.
Vaccinations: The safety of immunization with live-attenuated (live) vaccines with TZIELD-treated patients has not been studied. TZIELD may interfere with immune response to vaccination and decrease vaccine efficacy. Administer all age-appropriate vaccinations prior to starting TZIELD.
- Administer inactivated (killed) vaccines or mRNA vaccines at least 2 weeks prior to treatment. Inactivated vaccines are not recommended during treatment or 6 weeks after completion of treatment.
- Administer live vaccines at least 8 weeks prior to treatment. Live vaccines are not recommended during treatment, or up to 52 weeks after treatment.
ADVERSE REACTIONS
Most common adverse reactions were lymphopenia, vomiting, rash, leukopenia, diarrhea and headache.
USE IN SPECIFIC POPULATIONS
- Pregnancy: May cause fetal harm. To minimize exposure to a fetus, avoid use of TZIELD during pregnancy and at least 30 days prior to planned pregnancy. Report pregnancies to us at our Adverse Event reporting line at 1-800-633-1610 or visit https://ae.reporting.sanofi
- Lactation: A lactating woman may consider pumping and discarding breast milk during and for 20 days after TZIELD administration.
Please see full Prescribing Information, including Boxed WARNING and patient selection criteria.
INDICATION
TZIELD (teplizumab-mzwv) is indicated to delay the onset of Stage 3 type 1 diabetes (T1D) in adults and pediatric patients 1 year of age and older with Stage 2 T1D.

